Treatment with XIAFLEX
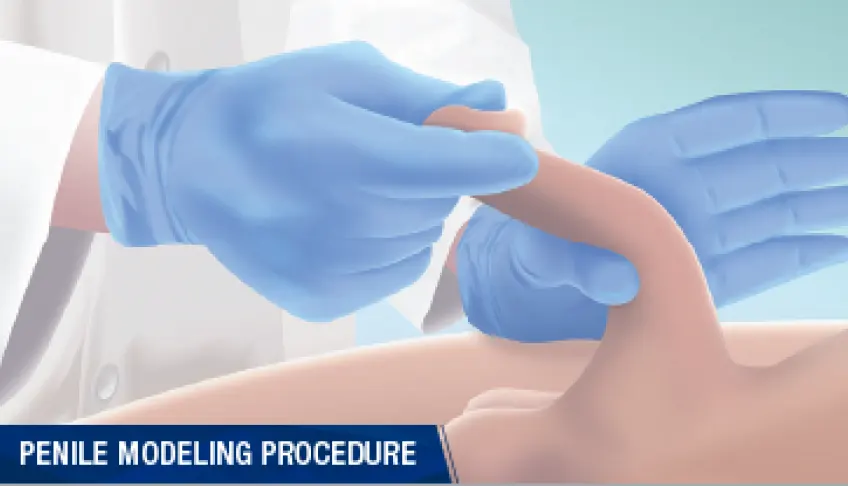
Procedure, expectations, and recommended guidelinesWatch an HCP-performed procedure
Take an in-depth look at the treatment procedure for XIAFLEX with one of the country’s

This menu will become active after the presentation of the BOXED WARNING.
Please see Important Safety Information throughout this video.
Dr Gelbard is a urologist with more than 30 years of experience in the study of Peyronie’s disease (PD). He has also served as the principal investigator in several studies and had been involved with the 2 pivotal clinical trials for XIAFLEX.
A treatment course of XIAFLEX takes ~24 weeks and consists of up to 4 treatment cycles approximately 6 weeks apart1
Treatment cycle (up to 4 cycles)
1–3 days
1–3 days
Approximately 6 weeks
If the curvature deformity is less than 15 degrees after the first, second, or third treatment cycle, or if further treatment is not clinically indicated, then subsequent treatment cycles should not be administered.1
The safety of more than 1 treatment course of XIAFLEX is not known.1
Each treatment cycle involves1:
- 3 office visits over the course of 3–7 days
- Each cycle begins with the injection of XIAFLEX into the palpable Peyronie’s plaque
- The second injection is performed 1–3 days after the first injection
- Based on patient’s penile swelling, you can assess if he is ready for the second injection
- In-office modeling is performed 1–3 days after the second injection
- The patient then performs at-home daily penile modeling for approximately 6 weeks
~80% of patients treated with XIAFLEX in Studies 1 and 2 received 8 injections over the course of 4 treatment cycles2,3

 Actor portrayal.
Actor portrayal.Penile modeling to help reduce erectile curvature and straighten the penile shaft1
By you in the office:
Perform in-office penile modeling procedure as described in the Prescribing Information during each treatment cycle 1 to 3 days after the second injection of XIAFLEX.
Helps to stretch and elongate the treated plaque1
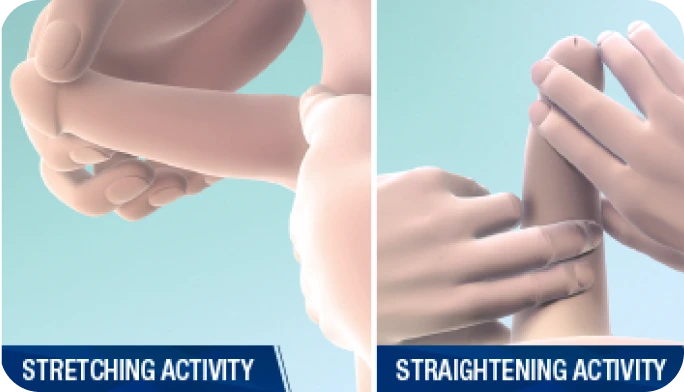
By patients at home:
Instruct patients on how to self-perform penile modeling activities at home each day for 6 weeks after each treatment cycle as described in the Medication Guide.1
Helps reduce erectile curvature1
AUA PD treatment guideline for XIAFLEX
In 2015, the American Urological Association (AUA) published a guideline that provides a clinical framework for the diagnosis and treatment of PD. The following guideline statements are specific to XIAFLEX.
Clinicians may administer intralesional collagenase clostridium histolyticum in combination with modeling by the clinician and by the patient for the reduction of penile curvature in patients with stable PD, penile curvature >30° and <90°, and intact erectile function (with or without the use of medications). (Moderate Recommendation; Evidence Strength Grade B)4
Clinicians should counsel patients with PD prior to beginning treatment with intralesional collagenase regarding potential occurrence of adverse events, including penile ecchymosis, swelling, pain, and corporal rupture. (Clinical Principle)4
Moderate Recommendations are directive statements that an action should (benefits outweigh risks/burdens) or should not (risks/burdens outweigh benefits) be undertaken because net benefit or net harm is moderate4
Body of Evidence Strength Grade B in support of a Moderate Recommendation indicates that the statement can be applied to most patients in most circumstances but that better evidence could change confidence4
A Clinical Principle is a statement about a component of clinical care that is widely agreed upon by urologists or other clinicians for which there may or may not be evidence in the medical literature.4
Important Safety Information
WARNING: CORPORAL RUPTURE (PENILE FRACTURE) OR OTHER SERIOUS PENILE INJURY IN THE TREATMENT OF PEYRONIE’S DISEASE
Corporal rupture (penile fracture) was reported as an adverse reaction in 5 of 1044 (0.5%) XIAFLEX‑treated patients in clinical studies. In other XIAFLEX‑treated patients (9 of 1044; 0.9%), a combination of penile ecchymoses or hematoma, sudden penile detumescence, and/or a penile "popping" sound or sensation was reported, and in these cases, a diagnosis of corporal rupture cannot be excluded. Severe penile hematoma was also reported as an adverse reaction in 39 of 1044 (3.7%) XIAFLEX‑treated patients.
Signs or symptoms that may reflect serious penile injury should be promptly evaluated to assess for corporal rupture or severe penile hematoma which may require surgical intervention.
Because of the risks of corporal rupture or other serious penile injury, XIAFLEX is available for the treatment of Peyronie’s disease only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the XIAFLEX REMS Program.
- Contraindications: XIAFLEX is contraindicated in the treatment of Peyronie’s plaques that involve the penile urethra due to potential risk to this structure and in patients with a history of hypersensitivity to XIAFLEX or to collagenase used in any other therapeutic application or application method
- Corporal Rupture or Other Serious Injury to the Penis: Injection of XIAFLEX into collagen-containing structures such as the corpora cavernosa of the penis may result in damage to those structures and possible injury such as corporal rupture (penile fracture). Therefore, XIAFLEX should be injected only into the Peyronie’s plaque and care should be taken to avoid injecting into the urethra, nerves, blood vessels, corpora cavernosa or other collagen-containing structures of the penis. Cases of localized skin and soft tissue necrosis occurring as sequelae of penile hematoma, some requiring surgical intervention, have been reported post-marketing
- Hypersensitivity Reactions, Including Anaphylaxis: In the double-blind, placebo-controlled portions of the clinical trials in Peyronie’s disease, a greater proportion of XIAFLEX-treated patients (4%) compared to placebo-treated patients (1%) had localized pruritus after up to 4 treatment cycles (involving up to 8 XIAFLEX injection procedures). The incidence of XIAFLEX-associated pruritus was similar after each injection regardless of the number of injections administered
- Because XIAFLEX contains foreign proteins, severe allergic reactions to XIAFLEX can occur. Anaphylaxis was reported in a post-marketing clinical trial in one patient who had previous exposure to XIAFLEX for the treatment of Dupuytren’s contracture. Healthcare providers should be prepared to address severe allergic reactions following XIAFLEX injections. The safety of more than one treatment course of XIAFLEX is not known
- Risk of Bleeding in Patients with Abnormal Coagulation: In the XIAFLEX controlled trials in Peyronie’s disease, 65.5% of XIAFLEX-treated patients developed penile hematoma, and 14.5% developed penile ecchymosis. Patients with abnormal coagulation (except for patients taking low-dose aspirin, eg, up to 150 mg per day) were excluded from participating in these studies. Therefore, the efficacy and safety of XIAFLEX in patients receiving anticoagulant medications (other than low-dose aspirin, eg, up to 150 mg per day) within 7 days prior to XIAFLEX administration is not known. In addition, it is recommended to avoid use of XIAFLEX in patients with coagulation disorders, including patients receiving concomitant anticoagulants (except for low-dose aspirin)
- Acute Post-Injection Back Pain Reactions: Post-marketing reports of acute lower back pain reactions, sometimes accompanied by radiation to the lower extremities, chest and arms, muscle spasms, chest pain, paresthesias, headache, and dyspnea, have been received by patients treated with XIAFLEX for Peyronie’s disease. These events can be mild to severe in intensity. The events typically lasted for 15 minutes and typically did not require intervention. Administer the smallest number of treatment cycles necessary to treat the patient’s curvature deformity
- Syncope and Presyncope: Most, but not all cases of syncope and presyncope in patients with Peyronie’s disease, occurred in association with post-injection penile pain and hematoma, penile pain with spontaneous erections, and pain during micturition. These potential triggers suggest a vasovagal mechanism. Make patients aware of the potential symptoms that could trigger syncope and presyncope after treatment with XIAFLEX.
If presyncopal symptoms occur, patients should remain recumbent until symptoms resolve. Syncope may be associated with bodily injuries, including concussion, head abrasion, and other accidental injuries
- In the XIAFLEX clinical trials for Peyronie’s disease, the most frequently reported adverse drug reactions (≥25%) and at an incidence greater than placebo included: penile hematoma, penile swelling, and penile pain.
- Acute post-injection lower back pain reactions have occurred in close temporal proximity to XIAFLEX treatments
- Cases of localized skin and soft tissue necrosis events as sequelae of penile hematoma, some of which required surgical intervention
- Syncope and presyncope have been reported in men treated with XIAFLEX for Peyronie’s disease. Most, but not all cases occurred in the immediate treatment period or within 1-2 days following injection. Bodily injuries associated with the syncopal events have been reported
References: 1. XIAFLEX® [package insert]. Rochester, MI: Endo USA, Inc. 2. Data on file. DOF-XPD-03. Endo USA, Inc.; January 7, 2016. 3. Data on file. DOF-XPD-04. Endo USA, Inc.; January 7, 2016. 4. Nehra A, Alterowitz R, Culkin DJ, et al. Peyronie’s disease: AUA guideline. J Urol. 2015;194(3):745-753.

