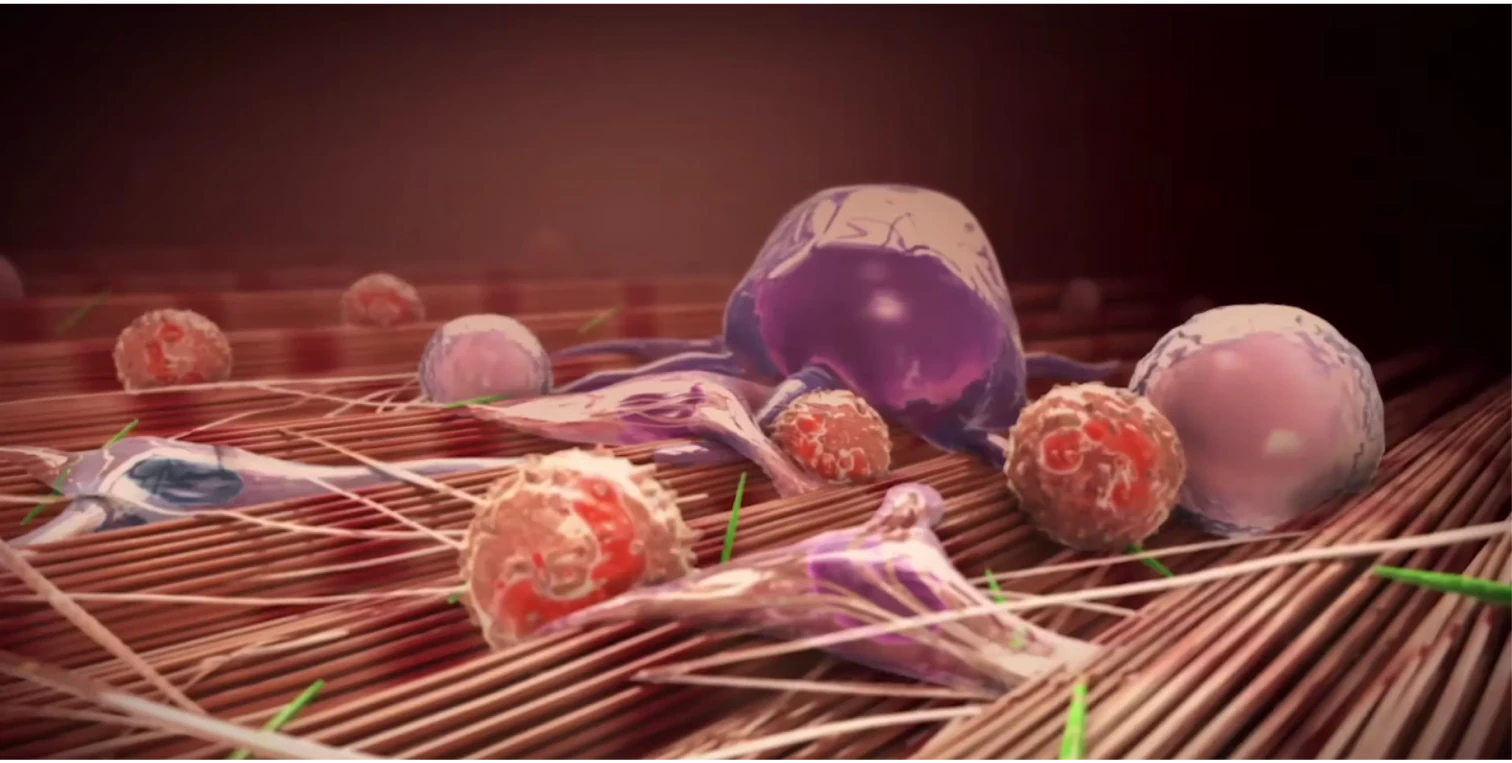
Mechanism of Peyronie’s disease (PD)
Watch and learn about the pathologic wound healing that is characteristic of the condition.
Treatment for adult men with Peyronie’s disease with a palpable plaque and curvature deformity of at least 30 degrees at the start of therapy
Watch a procedure performed by Dr Martin Gelbard.

This menu will become active after the presentation of the BOXED WARNING.
Please see Important Safety Information throughout this video.
Watch and learn about the pathologic wound healing that is characteristic of the condition.
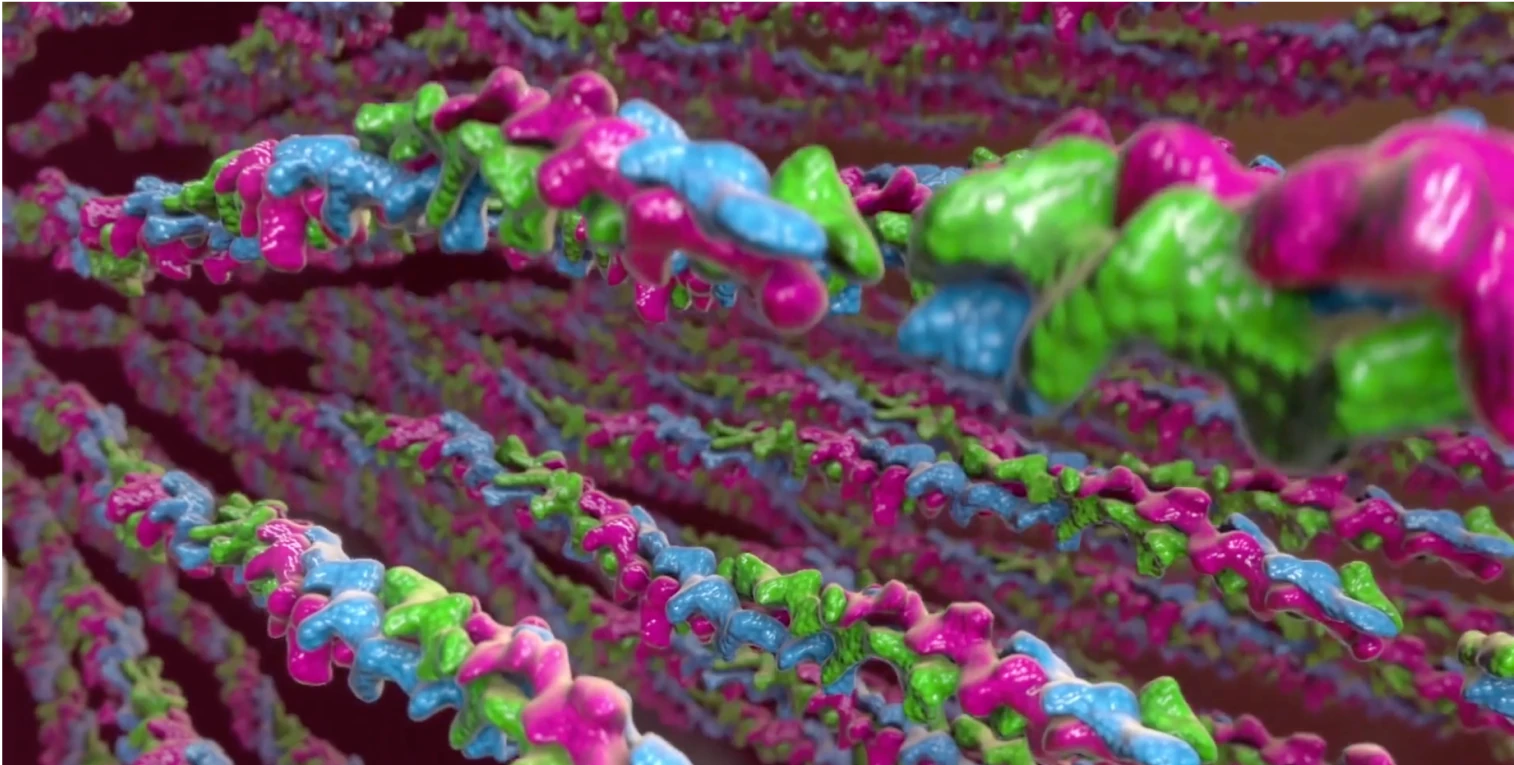
Watch XIAFLEX enzymatically disrupt collagen fibers to break down Peyronie’s plaques.
Watch four men talk about their PD diagnoses, and what happened next.

Watch real patients with PD consider their treatment options.

Watch real patients talk about their experiences with XIAFLEX.

HCPs/office staff can use this sheet to request assistance from Endo Advantage™.
Sample letter for you to reference when drafting your request for coverage of injections for treatment.
Have patients fill out this form to request copay assistance for XIAFLEX.
Have patients fill out this form to apply for the Patient Assistance Program.
Select the billing codes that most accurately describe the services provided.
Help patients get the conversation started using the helpful questions in this discussion guide.
Educate patients starting out with XIAFLEX on the 2 important parts of treatment: working with a Urology Specialist and at-home penile modeling.
WARNING: CORPORAL RUPTURE (PENILE FRACTURE) OR OTHER SERIOUS PENILE INJURY IN THE TREATMENT OF PEYRONIE’S DISEASE
Corporal rupture (penile fracture) was reported as an adverse reaction in 5 of 1044 (0.5%) XIAFLEX‑treated patients in clinical studies. In other XIAFLEX‑treated patients (9 of 1044; 0.9%), a combination of penile ecchymoses or hematoma, sudden penile detumescence, and/or a penile "popping" sound or sensation was reported, and in these cases, a diagnosis of corporal rupture cannot be excluded. Severe penile hematoma was also reported as an adverse reaction in 39 of 1044 (3.7%) XIAFLEX‑treated patients.
Signs or symptoms that may reflect serious penile injury should be promptly evaluated to assess for corporal rupture or severe penile hematoma which may require surgical intervention.
Because of the risks of corporal rupture or other serious penile injury, XIAFLEX is available for the treatment of Peyronie’s disease only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the XIAFLEX REMS Program.